The evasive nature of drug efficacy: implications for drug discovery. Probing the beta2 adrenoceptor binding site with catechol reveals differences in binding and activation by agonists and partial agonists. Kinetic evidence for intermediate conformational states. Sequential binding of agonists to the beta2 adrenoceptor. 
Agonist-induced conformational changes in the G-protein-coupling domain of the beta 2 adrenergic receptor. Caveolae and lipid rafts: G protein-coupled receptor signaling microdomains in cardiac myocytes. Roles of G-protein-coupled receptor dimerization. Regulation of GPCRs by endocytic membrane trafficking and its potential implications. Desensitization of G protein-coupled receptors. Beta-arrestin-mediated activation of MAPK by inverse agonists reveals distinct active conformations for G protein-coupled receptors. Beta-arrestin-dependent, G protein-independent ERK1/2 activation by the beta2 adrenergic receptor. Recent advances in cardiac beta(2)-adrenergic signal transduction. Inverse agonist activity of beta-adrenergic antagonists. Negative antagonists promote an inactive conformation of the beta 2-adrenergic receptor. Samama, P., Pei, G., Costa, T., Cotecchia, S. This paper provides a comprehensive analysis of sequence relationships between G-protein-coupled receptors in the human genome. Phylogenetic analysis, paralogon groups, and fingerprints. The G-protein-coupled receptors in the human genome form five main families. 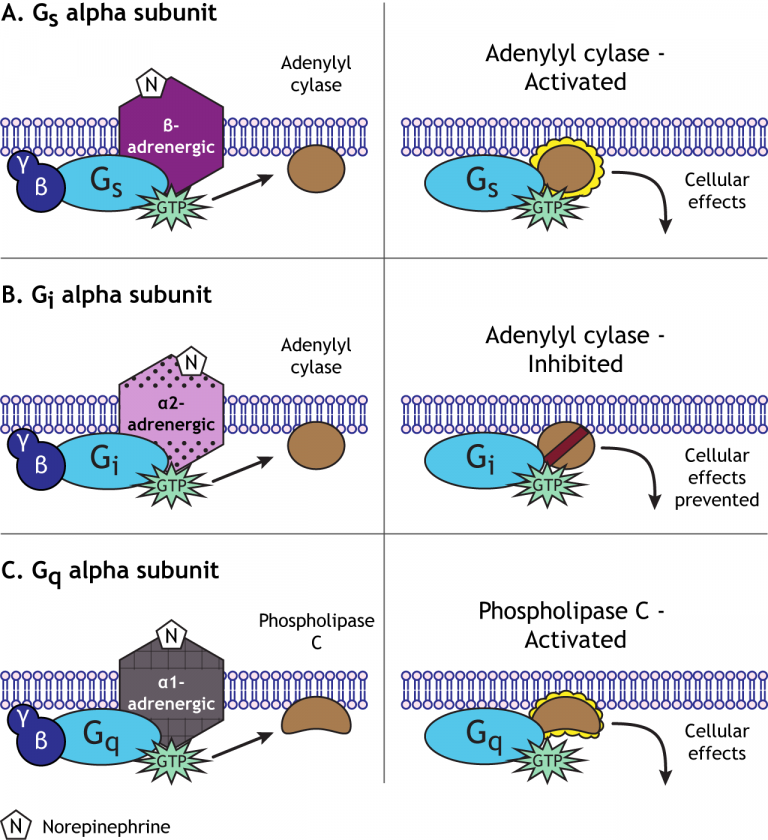
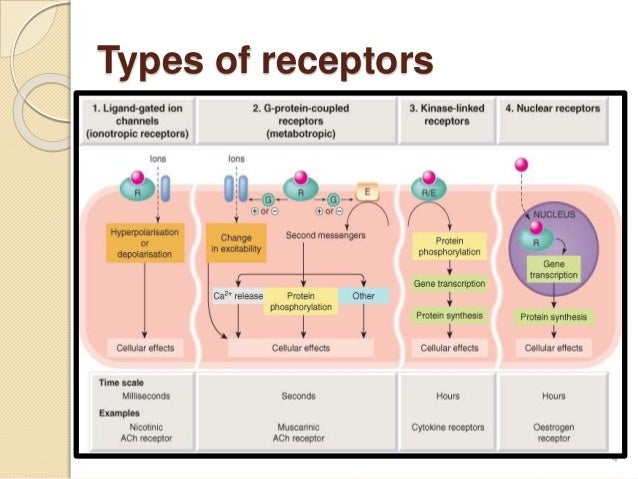
The pathways leading to PKC and PKA activation following the binding of a signal to a GPCR are summarized in Figure 8.4.12.Fredriksson, R., Lagerstrom, M. Like PKA, Protein kinase C phosphorylates a variety of proteins in the cell, altering their activity and thus changing the state of the cell. The increase in cytosolic calcium ion concentration has various effects, one of which is to activate a protein kinase called protein kinase C (C for calcium), together with the DAG made in the earlier step. This causes calcium channels in the ER membrane to open and release large amounts of calcium into the cytoplasm from the ER lumen, as shown in the figure below. First, IP3 diffuses to the endoplasmic reticulum membrane where it binds to gated calcium ion channels. The IP3 and DAG produced by activated phospholipase C work together to activate a protein kinase.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
